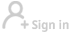Moderna plans to ask regulators to expand its COVID-19 vaccine's use to 12 to 17-year-olds

WASHINGTON - Moderna, Inc. said on Tuesday that its COVID-19 vaccine was effective in children age 12 to 17 years old in a new study, and is planning to ask health regulators in the United States and other countries early next month to expand the shot's use to this age bracket.
The Cambridge, Massachusetts, company said that the vaccine induced immune responses among children that were comparable to those seen in a study of adults last year, a finding that could clear the way for a second shot for use in adolescents after Pfizer-BioNTech.
There were no cases of symptomatic COVID-19 among vaccine recipients, Moderna said, suggesting 100 percent vaccine efficacy in adolescents, though overall very few among the 3,700 children in the study got sick.
Based on the results, Moderna said that it plans in early June to request that regulators in the United States and other countries authorize the use of its vaccine in children age 12 to 17 years old.
The US Food and Drug Administration (FDA) could make a decision within weeks of the request if it follows the same kind of timetable if took with Pfizer Inc.'s PFE shot. The agency took about a month to clear Pfizer's request for use of its COVID-19 vaccine in adolescents, said The Wall Street Journal on Tuesday.
Immunizing children is crucial to developing the communitywide immunity needed to move fully past pandemic precautions, health specialists were quoted as saying.
Proof of the vaccines' efficacy and safety for adolescents is helping school officials and other leaders as they plan for the fall. Mayor Bill de Blasio has said that all public school students in New York City, the largest school system in the United States, would return to in-person learning in the fall, The New York Times reported on Tuesday.
In December, the FDA authorized use of Moderna's shot in people 18 years and older, and the COVID-19 vaccine from Pfizer and its partner BioNTech SE was cleared for 16 and 17 years old, along with adults. Earlier this month, the FDA authorized the Pfizer shot for use in children age 12 to 15 years old.